If you’ve been in fitness, recovery, or longevity circles lately, you’ve probably heard of BPC-157—the peptide everyone calls the “Wolverine compound” because people swear it speeds up healing in tendons, ligaments, muscles, and even the gut. The nickname is pretty spot-on—it really does feel like something out of a comic book when recovery happens faster than expected.
Discovery and Origins of BPC-157
The story of BPC-157 begins in the early 1990s in Croatia, at the University of Zagreb’s School of Medicine. Pharmacologist Predrag Sikirić and his research team were investigating protective factors in human gastric juice—substances that help shield the stomach lining from damage. In their foundational work, published starting in 1993 (notably in the Journal of Physiology-Paris and related papers), they isolated a larger “body protection compound” (BPC) from gastric secretions and identified a stable, active 15-amino-acid fragment as the key player.
This fragment—now known as BPC-157—showed remarkable cytoprotective (cell-protecting) effects in early animal models, with no close structural similarity to other known peptides at the time. Sikirić’s group went on to publish extensively on its potential, building a body of preclinical research that still forms the backbone of what we know today. It’s a classic case of serendipitous discovery: looking for something to protect the gut, they uncovered a molecule with surprisingly broad applications.
What Exactly Is BPC-157?
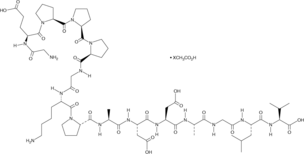
BPC-157 is a synthetic 15-amino-acid peptide derived from a natural protective fragment in human gastric juice, but engineered for stability and research use. Its amino acid sequence is:
Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val
It supports tissue repair by promoting angiogenesis, modulating growth factors, reducing inflammation, and protecting against damage. A key advantage: it’s stable in gastric acid, enabling potential oral dosing—unlike most peptides that degrade quickly.
Half-Life & Clearance
BPC-157 has a very short half-life—typically under 30 minutes in animal studies. It is rapidly broken down in the liver and cleared through the kidneys. Because of this fast clearance, most protocols recommend daily dosing, often split into two injections (morning and evening) to maintain steadier exposure.
Preclinical studies (primarily in rodents) show faster healing of tendons, ligaments, muscles, bones, and gastrointestinal tissues. A 2024 systematic review confirmed improved functional, structural, and biomechanical outcomes in injury models, with no major toxicity observed. Human data is still limited—no big randomized trials yet as of early 2026—but early pilots and reviews call it promising, especially for GI issues.
How BPC-157 Is Manufactured
BPC-157 is fully synthetic and produced entirely in the lab—no extraction from human or animal tissue. The active 15-amino-acid fragment is made using solid-phase peptide synthesis (SPPS), the standard method for short peptides.
🔬 The SPPS Process
- The chain starts on a solid resin support with the C-terminal amino acid (Valine) attached
- Protected amino acids are added one by one with coupling agents (e.g., HBTU or HATU)
- The cycle (deprotect, couple, wash) repeats until the full sequence is built
- The peptide is then cleaved, deprotected, purified by HPLC, and freeze-dried into powder (usually acetate or arginate salt)
This chemical process delivers high purity when done correctly. Quality varies widely—reputable labs and compounding pharmacies produce clean product, but unregulated sources can contain impurities or incomplete chains.
Key Studies: Preclinical and Human Pilot
Animal Study
Staresinic et al. (2003) – Achilles Tendon Healing in Rats
Published in the Journal of Orthopaedic Research, this study examined BPC-157 in rats with sharply transected Achilles tendons (5 mm defect). Daily BPC-157 doses led to significantly better outcomes vs. saline controls: higher Achilles Functional Index throughout 14 days, increased biomechanical strength, superior microscopic features (more fibroblasts, better collagen organization), and macroscopically smaller defects with eventual full tendon integrity restoration. In vitro, it protected tendon fibroblasts from oxidative damage.
Note: Preclinical only—direct human translation is limited.
Human Pilot
Lee et al. (2024) – Interstitial Cystitis Pilot Study
Published in Alternative Therapies in Health and Medicine, this small retrospective pilot involved 12 women with moderate-to-severe interstitial cystitis refractory to pentosan polysulfate (the only FDA-approved treatment). Patients received intravesical injections of compounded BPC-157 (total 10 mg). At 6 weeks follow-up, 10 of 12 reported complete symptom resolution, with the remaining two noting ~80% improvement. No adverse events were reported.
Limitations: Small sample, open-label, no placebo control, retrospective chart review, no long-term data. Preliminary at best.
These examples underscore the evidence gap: repeatable, multi-faceted animal effects versus sparse, uncontrolled human pilots.
Reported Benefits and the Anecdotal Landscape
From user reports across forums and clinic feedback, common applications include:
- Accelerated recovery from tendon/ligament strains or tears
- Reduced joint inflammation and pain
- Support for muscle repair post-injury
- Gastrointestinal healing (e.g., leaky gut, ulcers, IBD models in animals)
Anecdotes range from “life-changing” (e.g., returning to sport weeks ahead of schedule) to “meh, didn’t notice much.” Variability likely stems from sourcing quality, individual response, concurrent rehab, and the ever-present placebo factor.
Typical Protocols
Without official guidelines, protocols draw from preclinical dosing scaled to humans and clinical practitioner experience:
| Method | Dose / Notes |
|---|---|
| Standard SubQ | 250–500 mcg/day, 1–2 injections |
| Localized Injury | Inject near site (e.g., knee, shoulder) |
| Systemic | Abdominal subQ works well |
| Oral (gut use) | 200–500 mcg daily — stable in gastric acid |
| Cycle Length | 4–6 weeks on, equal time off |
| Weight-based | ~2–4 mcg/kg/day (most stick to flat 250–500 mcg) |
These are not prescriptions—just patterns observed in telehealth settings and user reports. Always reconstitute properly, store refrigerated, and use sterile technique.
Reconstitution Guide
Use bacteriostatic water (BAC water) for multi-dose stability. Add water slowly down the side of the vial (not directly onto the powder to avoid foaming), gently swirl until fully dissolved—do not shake. Store refrigerated and use within 4–6 weeks.
💉 Quick Reference: 5 mg Vial
- Add 2 mL BAC water → 2,500 mcg/mL
- 10 units (0.1 mL) = 250 mcg
- 20 units (0.2 mL) = 500 mcg
💉 Quick Reference: 10 mg Vial
- Add 2 mL BAC water → 5,000 mcg/mL
- 5 units (0.05 mL) = 250 mcg
- 10 units (0.1 mL) = 500 mcg
Safety Profile and Cautions
Preclinical studies show a favorable safety profile—no major organ toxicity observed across multiple systems. In the few small human pilots, tolerability has been good, with only mild side effects reported: injection-site irritation, occasional transient headache, or nausea.
Significant unknowns remain:
- Long-term effects (no extended human data exists)
- Potential immunogenicity (immune response to the peptide itself)
- Theoretical angiogenesis risk (could promote growth in undetected tumors)
- Purity and contamination risks from unregulated sources
The FDA’s Category 2 classification for compounding is a clear signal: meaningful safety and characterization gaps persist.
Final Thoughts
BPC-157 stands out in the peptide space for its preclinical healing research, much of it built on Sikirić’s early work. The animal studies show consistent patterns, and the limited human pilots add some encouraging signals. Honestly, I absolutely love this peptide.
What really catches my attention is how many people report strong results: faster recovery from tendon/ligament issues, quicker return after workouts or sports, reduced joint pain, and better gut function. A lot of them describe getting back to normal activity sooner than expected, often after being sidelined for months. I’ve also seen several stories where doctors were surprised—sometimes shocked—at how fast patients healed from surgeries, with earlier mobility and less scar tissue than usual.
If that kind of outcome sounds like it could help with a lingering injury, talking to a clinician familiar with peptides might be worth considering. The real-world feedback keeps it relevant.
What’s Your Experience with BPC-157?
Has it made a noticeable difference in your recovery or training? Share your experience in the comments—this community is full of useful insights, and the field moves quickly. 👇
